Connecting Israel’s Tech Ecosystem
Fostering the Inclusive Growth, Resilience and Global Competitiveness of Israel’s Advanced Technology Industries
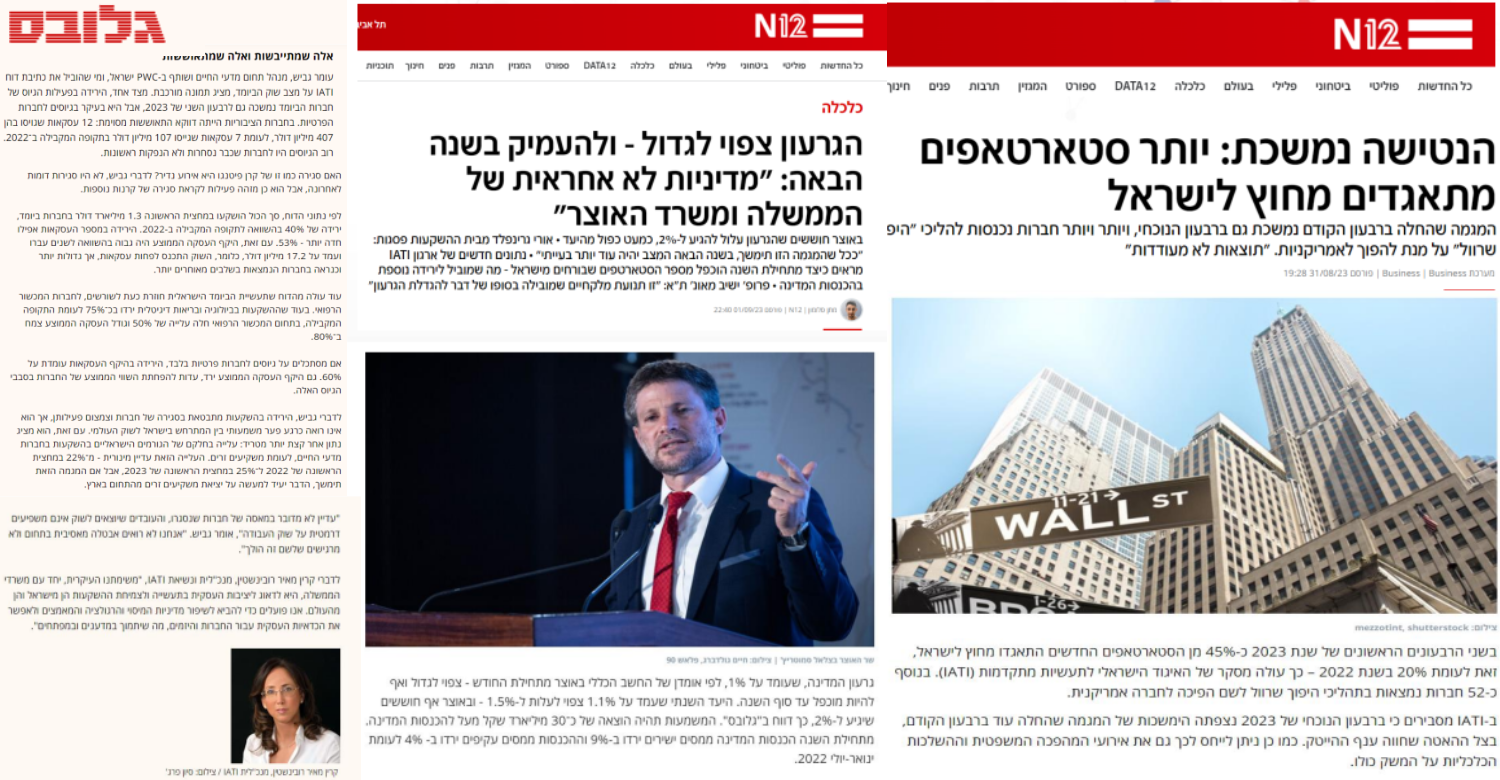
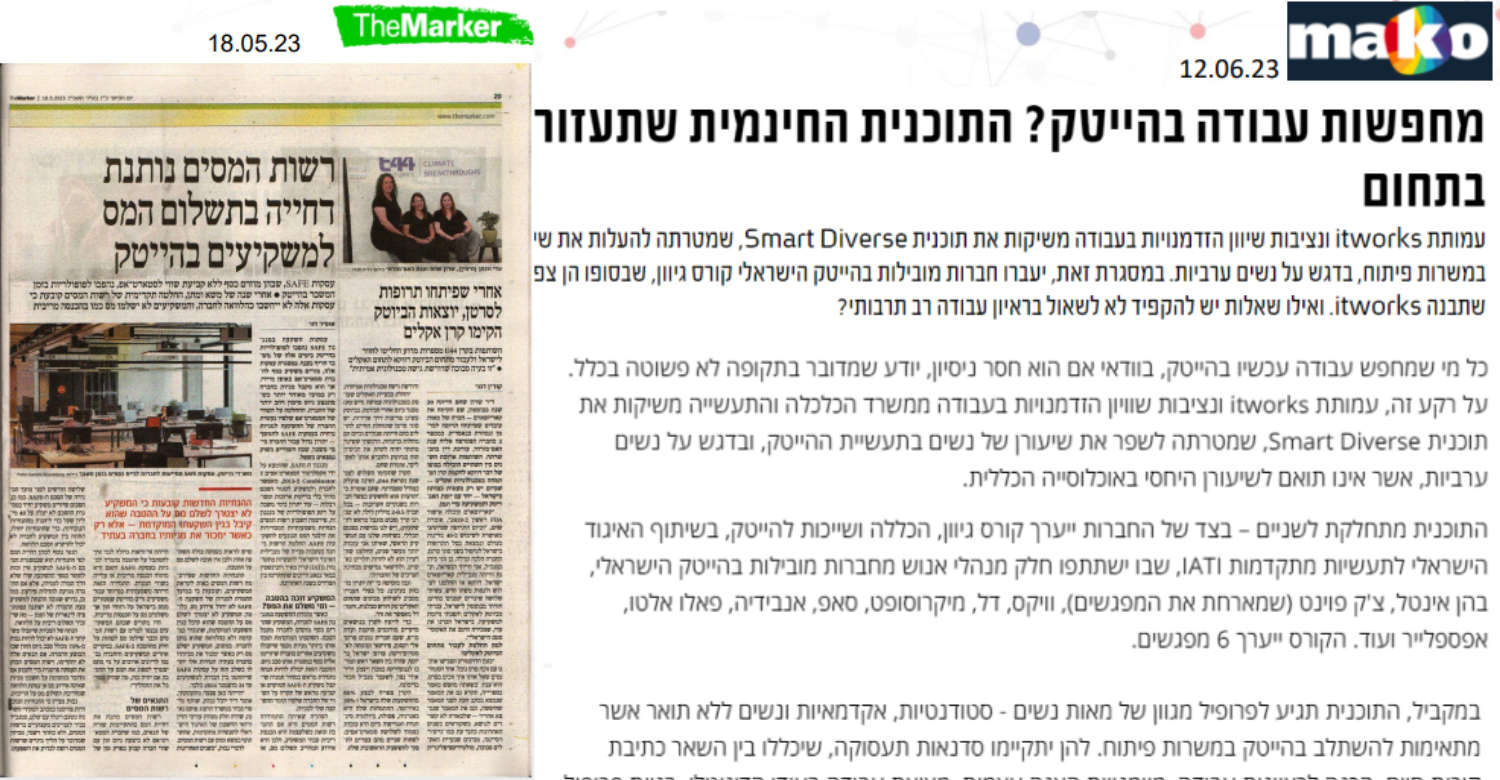
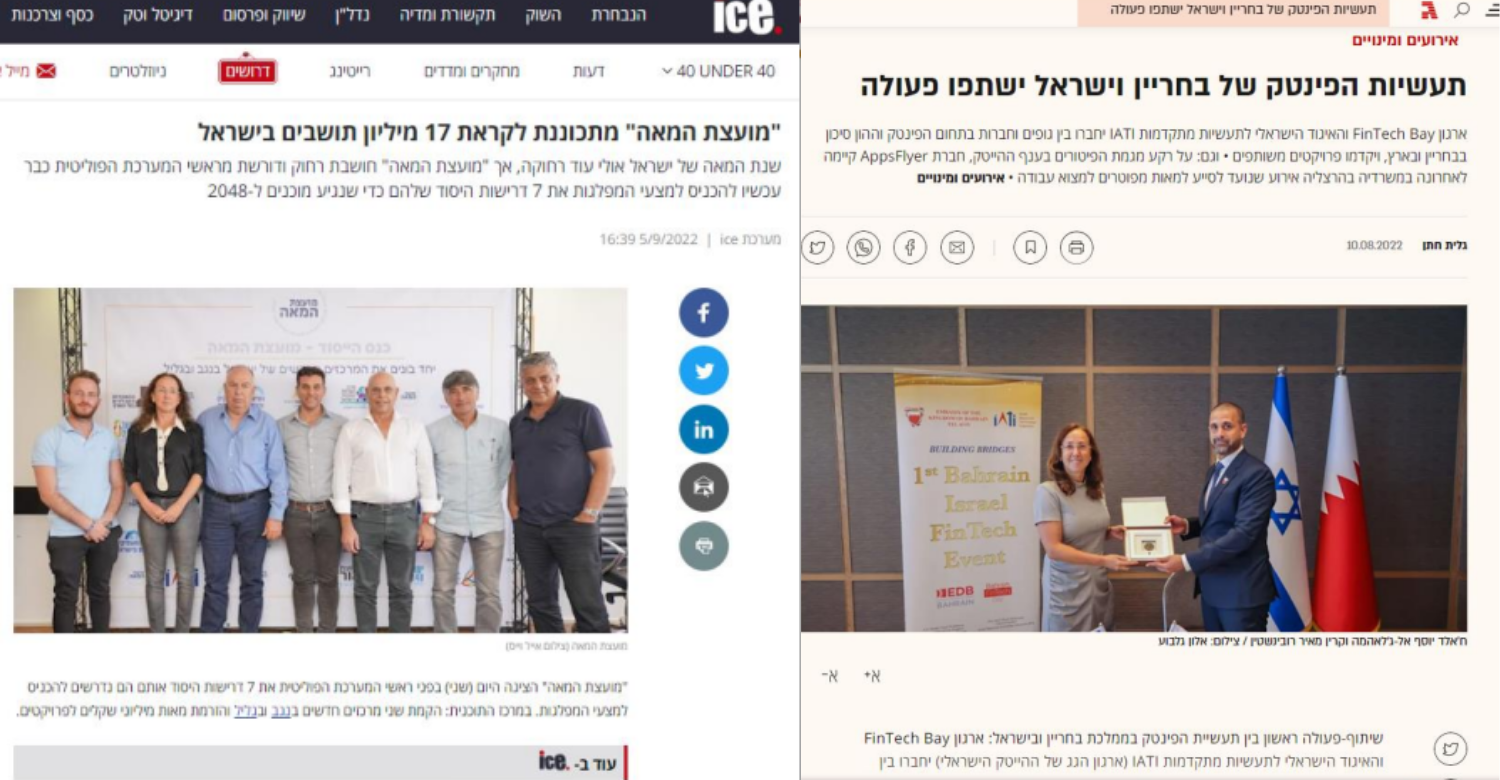
Activity Highlights
About us
A nonprofit association of Israel’s advanced tech industry players, IATI is on a mission to foster business certainty and stability for advancing Israel’s high-tech and health-tech industries.
This, in turn, furthers national resilience and inclusive economic growth and strengthens Israel’s position as a global innovation leader.
Media
Stay Connected
Whether you’re an IATI member, an Israeli tech industry player looking to join us, or a global stakeholder seeking the latest voice or report from Israel’s tech industry – we want to hear from you.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Terms and Conditions & Privacy Policy
Terms of Use
Website Terms of Use:
“IATI” means Israel’s Association of Tech Industries Association (R.A. number 580554079 existing under the laws of the State of Israel) with offices at Medinat Ha’Yehudim 89, Hertzliya Pituach, Israel .
PLEASE READ THESE TERMS OF USE (the “Terms of Use”) CAREFULLY BEFORE USING THE IATI WEBSITE.
Accepting the Terms of Use
By accessing and using the IATI website (the “Website”) or by clicking to accept or agree to the Terms of Use where this option is available, you accept and undertake these Terms of Use and you signify your agreement to and intent to be bound by these Terms of Use. These Terms of Use apply to all users of the Website.
You may not use the Website or any information contained herein if you do not agree to the Terms of Use or if you are not permitted to accept the Terms of Use under any applicable law, regulation or obligation, including, without limitation, if you are not of legal age to form a binding contract.
Website Access
IATI hereby grants you permission to use the Website as set forth in these Terms of Use, provided that: (i) comply in full with these Terms of Use (ii) your use of the Website as permitted is solely for your personal, noncommercial use; and (ii) you will not copy or distribute any part of the Website in any medium without IATI’s prior written authorization; and (iii) you will not access the Website using a different interface than the interface provided by IATI, including, without limitation, by means of data mining, robots, or similar data gathering and extraction tools; and (iv) You will not to circumvent, disable, disrupt or otherwise interfere with any feature of the Website inclusive of it’s servers or networks; and (v) you will not alter or modify any part of the Website other than as may be reasonably necessary to use the Website for its intended purpose; and (vi) you will otherwise comply with any applicable law and with of these Terms of Use; and (vii) you will not upload, transmit or otherwise make available any content that is unlawful, harmful, abusive, defamatory, vulgar, libelous, invasive of another’s privacy, hateful, or racially, ethnically or otherwise objectionable or content that you do not have the right to upload, transmit, or make available.
In order to access some features of the Website, you will have to create an account. By creating your account, you agree to provide true, accurate, current and complete information about yourself as prompted by our registration processes, and to maintain and update your information, as needed. You may not use another users account without obtaining prior written permission. You shall not impersonate any person or entity or misrepresent your identity or affiliation with any person or entity, including using another person’s username, password or other account information, or another person’s name, likeness, voice, image or photograph.
You are solely responsible for maintaining the confidentiality of your account, password or other account information and any activity that is occurs on your account, and you must keep your account password secure. You must notify IATI immediately of any breach of security or unauthorized use of your account. You will be liable for any use of your account or password, including without limitation, our losses due to any unauthorized use of your account. We will not be liable for any losses incurred by you or anyone on your behalf and caused due to any unauthorized use of your account.
Content and Rights
The content of the Website, including, without limitation, text, software, scripts, graphics, photos, sounds, music, videos, animations, designs, user interface (“Content”) and the trademarks, service marks and logos contained therein (“Marks”), are owned or licensed in by IATI, and you hereby agree not to use, copy, reproduce, distribute, transmit, broadcast, display, sell, license, or otherwise exploit them for any other purposes whatsoever without the prior written approval by IATI and its licensors. IATI reserves all rights not expressly granted in and to the Website and the Content.
IATI distributes and publishes content supplied by third parties. IATI has no control over such content and, without derogating from Section 4 below, IATI assumes no warranty with respect to such content.
Some sections of the Website may provide you and other visitors of the Website with the opportunity to submit, post, display, transmit and/or exchange information, ideas, opinions, photographs, images, video, sound recordings, , creative works or other information, messages, transmissions or materials, including your name, to IATI (collectively: “Submission”). You retain your rights in content which you submit, post or display on the Website. By submitting, posting or displaying such content you represent and warrant that it complies with these Terms of Use, and you acknowledge and agree that you are granting IATI a perpetual, worldwide, unlimited, royalty-free, irrevocable, transferable (in whole or part), sublicensable and non-exclusive license to use, distribute, publicly display, reproduce, copy, edit, adapt, modify, translate, publish and publicly perform any such content. You represent and warrant to IATI that you have all the rights, power and authority necessary to grant the above license.
You grant the above mentioned license to IATI whether or not your submission (or any part thereof) is actually posted on the Website or used by IATI. You acknowledge and agree that IATI may, at its sole discretion, elect to post or not post your submission, or to remove your submission from the IATI Website after it has been posted or published, at any time, for any reason and without notice, and not to use, exhibit or otherwise exploit your submission in any manner whatsoever. You further acknowledge and agree that you post, submit or upload your submission voluntarily, and not in confidence, and that no confidential relationship is intended or created between IATI, and/or any other person or entity, on the one hand, and you, on the other hand, by your submission.
You are solely responsible for your Submission and any consequences of posting or publishing it on the Website in any form. These sections of the WebStite are public and not private, and you acknowledge and agree that you have no expectation of privacy with respect to any Submission posted or publisher in any such section of the Website.
No Warranty
THE WEBSITE AND ITS CONTENT ARE PROVIDED ON AN “AS-IS” AND “AS-AVAILABLE” BASIS. YOUR USE OF THE WEBSITE IS ON YOUR OWN RISK. TO THE FULLEST EXTENT PERMITTED BY LAW, IATI, ITS OFFICERS, DIRECTORS, EMPLOYEES, AND AGENTS DISCLAIM ALL WARRANTIES, EXPRESS OR IMPLIED, IN CONNECTION WITH THE WEBSITE AND YOUR USE THEREOF. IATI MAKES NO WARRANTIES OR REPRESENTATIONS ABOUT THE ACCURACY OR COMPLETENESS OF THE WEBSITE’S CONTENT AND ASSUMES NO LIABILITY OR RESPONSIBILITY FOR ANY (I) ERRORS, MISTAKES, OMISSIONS OR INACCURACIES IN THE WEBSITE OR ANY RELIANCE ON THE CONTENT, (III) ANY UNAUTHORIZED ACCESS TO OR USE OF OUR SECURE SERVERS AND/OR ANY AND ALL PERSONAL INFORMATION AND/OR FINANCIAL INFORMATION STORED THEREIN, (IV) ANY CHANGE, INTERRUPTION OR CESSATION OF THE WEBSITE, TEMPORARY OR PERMANENT, (V) ANY BUGS, VIRUSES, TROJAN HORSES, OR THE LIKE WHICH MAY BE TRANSMITTED TO OR THROUGH OUR WEBSITE BY ANY THIRD PARTY. IATI DOES NOT WARRANT, ENDORSE, GUARANTEE, OR ASSUME RESPONSIBILITY FOR ANY PRODUCT OR SERVICE ADVERTISED OR OFFERED BY A THIRD PARTY THROUGH THE IATI WEBSITE OR ANY LINKED WEBSITE OR FEATURED IN ANY BANNER OR OTHER ADVERTISING OR ANY USER SUBMMITION FEATURED IN THE WEBSITE, AND IATI WILL NOT BE A PARTY TO OR IN ANY WAY BE RESPONSIBLE FOR MONITORING ANY TRANSACTION BETWEEN YOU AND THIRD-PARTY PROVIDERS OF PRODUCTS OR SERVICES. YOU ASSUME THE TOTAL RESPONSIBILITY FOR YOUR USE OF THE WEBSITE, SERVICES, CONTENT AND ANY WEBSITE LINKED THERETO. YOUR SOLE REMEDY AGAINST IATI FOR DISSATISFACTION WITH THE WEBSITE, SERVICES AND/OR CONTENT IS TO STOP USING THE WEBSITE, SERVICES OR ANY SUCH CONTENT.
Limitation of Liability
IN NO EVENT SHALL IATI, ITS OFFICERS, DIRECTORS, EMPLOYEES, OR AGENTS, BE LIABLE TO YOU FOR ANY INDIRECT, INCIDENTAL, SPECIAL, PUNITIVE, OR CONSEQUENTIAL DAMAGES, WHETHER BASED ON WARRANTY, CONTRACT, TORT, OR ANY OTHER LEGAL THEORY, AND WHETHER ORNOT THE COMPANY IS ADVISED OF THE POSSIBILITY OF SUCH DAMAGES. THE FOREGOING LIMITATION OF LIABILITY SHALL APPLY TO THE FULLEST EXTENT PERMITTED BY LAW IN THE APPLICABLE JURISDICTION.
IATI SHALL NOT BE LIABLE FOR USER SUBMISSIONS OR THE DEFAMATORY, OFFENSIVE, OR ILLEGAL CONDUCT OF ANY THIRD PARTY AND THAT THE RISK OF HARM OR DAMAGE FROM THE FOREGOING RESTS ENTIRELY WITH YOU.
The Website is controlled and offered by IATI from its facilities Israel. IATI makes no representations that the IATI Website is appropriate or available for use in other locations. Those who access or use the IATI Website from other jurisdictions do so at their own volition and are responsible for compliance with local law.
Assignment
These Terms of Use, and any rights and licenses granted hereunder, may not be transferred or assigned by you, but may be assigned by IATI without restriction.
Privacy Policy
IATI only collects information that IATI believes will improve its services. Like the operators of most websites, IATI uses analytic and reporting technologies to record Non-Personal Information. IATI will not provide your personal information to any third parties without your consent or as required by a competent authority. IATI takes all reasonable precautions to prevent the loss, misuse or alteration of your personal information. However, data transmission over the internet is inherently insecure, and we cannot guarantee the security of your personal data. The Website contains external links to other websites and we are not responsible for the privacy policies of third party websites.
Copyright Protection
IATI is committed to protecting intellectual property rights. However, IATI cannot – and does not – monitor all of the content on its website. If any of your rights were infringed by IATI Website please let us know immediately. Our designated agent for notice of alleged copyright infringement on the Website is:
IATI
Electra Bldg B, 1st floor
P.O. Box 12591
Abba Eban 8
Hertzliya Pituach [email protected]
A notification of a claimed infringement must be in a written form, addressed to the designated agent as set forth above, and must include substantially all of the following:
A physical or electronic signature of the owner (or person authorized to act on behalf of the owner) of an exclusive right that is allegedly infringed;
Specific identification of the copyrighted work claimed to have been infringed, or if multiple copyrighted works are covered by a single notification, a list of each copyrighted work claimed to have been infringed;
Information related to the work(s) reasonably sufficient for IATI to promptly locate the work (e.g. title of work, location within the Website, etc.);
Information reasonably sufficient to permit IATI to directly contact the complaining party, such as a complete name and address, telephone number, and an email address;
A statement that the complaining party has a good faith belief that use of the work(s) in the manner complained of is not authorized by the copyright owner, its agent, or the law;
A statement requesting that IATI take a specific act with respect to the alleged infringement (e.g., removal, access restricted or disabled); and
A statement that the information in the notification is accurate, and under penalty of perjury, that the complaining party is authorized to act on behalf of the owner of an exclusive right that is allegedly infringed. In the event your copyright infringement notice is discovered to be false, IATI reserves its rights to take any legal measure it is entitled to under any applicable law.
Purchase of Products and Payments
Subject to any applicable law, you may place an order for a product, service or association membership fee through the Website only if you are at least 18 years old. All such transactions at the website are secured according to PCI international standard by Net Pay Ltd., IATI’s payment service provider. Payment becomes due and owing immediately after the order is placed. Our cancellation policy for orders of products, services or association membership fee is subject to the Israeli Consumer Protection Act, 1981, except in transactions where section 14c(d) of the act is applicable.
Association membership will take effect upon completion of payment in accordance with the IATI Articles of Association and subject thereto.
Without derogating from Sections 4 and 5, IATI will not be liable for any failure, delay, expense or loss incurred by you which is beyond IATI’s reasonable control.
General
The IATI Website shall be deemed solely based in Israel and shall be deemed a passive website that does not give rise to personal jurisdiction over IATI, either specific or general, in jurisdictions other than Israel. These Terms of Use shall be governed by the laws of the State of Israel. Any claim or dispute between you and IATI that arises in whole or in part from the Website shall be decided exclusively by a court of competent jurisdiction located Tel-Aviv, Israel. If any provision of these Terms of Use is deemed invalid by a court of competent jurisdiction, the invalidity of such provision shall not affect the validity of the remaining provisions of these Terms of Use, which shall remain in full force and effect. No waiver of any term of this these Terms of Use shall be deemed a further or continuing waiver of such term or any other term, and IATI’s failure to assert any right or provision under these Terms of Use shall not constitute a waiver of such right or provision. With respect to any person other than registered users, IATI reserves the right to amend these Terms of Use at any time and without notice, and it is your responsibility to review these Terms of Use for any changes. All correspondence, negotiations and proceedings related to this Agreement will be in the English language only.
Privacy Policy
IATI only collects information that IATI believes will improve its services. Like the operators of most websites, IATI uses analytic and reporting technologies to record Non-Personal Information. IATI will not provide your personal information to any third parties without your consent or as required by a competent authority. IATI takes all reasonable precautions to prevent the loss, misuse or alteration of your personal information. However, data transmission over the internet is inherently insecure, and we cannot guarantee the security of your personal data. The Website contains external links to other websites and we are not responsible for the privacy policies of third party websites.